Content
- Composition and form of release
- Terms of sale, prices
- Pharmacological properties
- Enalapril
- Hydrochlorothiazide
- Combination of both substances
- Indications for use
- Contraindications
- Method of administration and dosage
- Arterial hypertension
- Pretreatment with diuretics
- Kidney function disorders
- Elderly people
- Side effects
- Overdose
- special instructions
- Drug interactions
- Analogs
- Storage conditions
- Video about the action of the drug Co-Rentitek
The combined drug Koreitek possesses diuretic and antihypertensive action. The indication for the appointment is the treatment of mild to moderate hypertension. Also, the medication is recommended for use in patients whose condition is stabilized by taking the active ingredients included in the medication separately in similar dosages.
Composition and form of release
The drug is produced in the form of round, yellow, bicuspid tablets with a corrugated edge. On one half there is an embossing in the form of the inscription "MSD 718" on the other - at risk.
7 tablets in a blister, or 56 tablets together with 1 or 2 plastic cases in a high-density bottle.
Cardboard packages contain 2 or 4 blisters (14 and 28 tablets each, respectively), or 1 bottle.
Active ingredients (mg in 1 tab.):
- enalapril maleate - 20;
- hydrochlorothiazide - 12.5.
Koreitek
Additional components:
- corn starch;
- sodium bicarbonate;
- iron oxide pigment yellow;
- magnesium stearate;
- lactose monohydrate;
- pregelatinized corn starch.
Terms of sale, prices
Korenitek can only be purchased with a prescription from the treating doctor, which is an indication for use. The price varies on the number of tablets in the package and the pharmacy network in which the drug is sold.
Pharmacological properties
Korenitek (indications for use are due to medicinal properties, which are aimed at lowering blood pressure, the content of sodium ions in the vessel wall, OPSS, as well as an increase in urine output) has an effect that is observed day. From this it follows that the medication is prescribed for hypertension. Therapy with this medication is more effective than taking its active substances separately from each other.
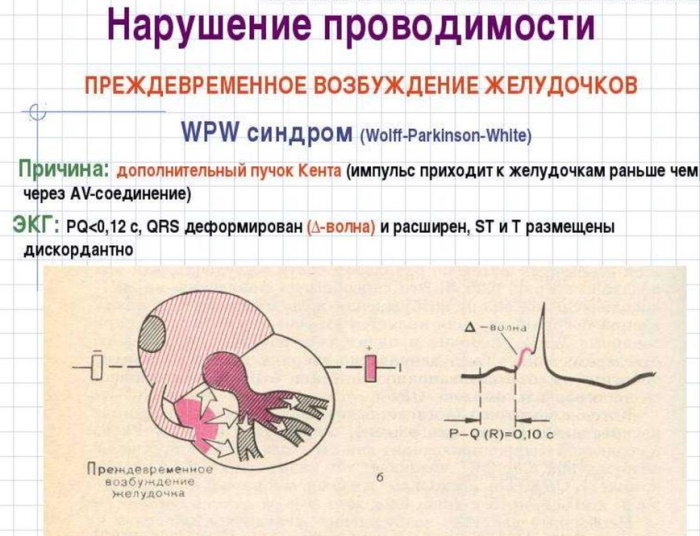
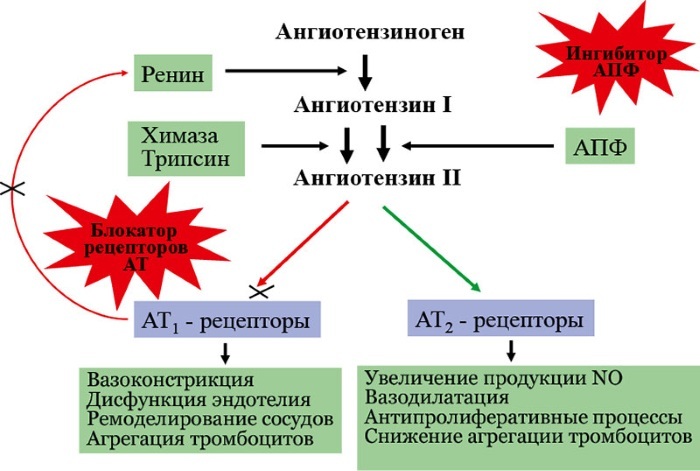
Enalapril is an ACE inhibitor. As it is absorbed, it is transformed into enalaprilat, the action of which is to reduce blood levels of angiotensin II, which stimulates the activation of plasma renin and a decrease in excretion aldosterone. The substance also prevents the destruction of bradykin.
A decrease in the total parasympathetic resistance of the arteries and a certain increase in the contractions of the heart muscle contribute to a decrease in blood pressure. The drug increases blood flow in the kidneys, while not changing the filtration rate, provided that it was not initially lowered in the patient.
Hydrochlorothiazide is an antihypertensive and diuretic that stimulates renin activation. In combination with enalapril, a more significant decrease in blood pressure is observed. Its sharp increase with drug withdrawal is not observed.
The maximum effect after administration is achieved after 2 hours. The pressure decreases after 1 hour. The duration of action is dependent on the dosage.
Enalapril
The substance provokes a decrease in blood pressure without a significant increase in the frequency of contractions of the heart muscle. Orthostatic symptomatic hypotension is rare. In some cases, several weeks must pass from the start of treatment in order to achieve the best reduction in blood pressure. The end of therapy with this substance does not provoke a sudden rise in blood pressure.
Inhibition of ACE activity is observed after a single oral administration after 2-4 hours. The pressure decrease indicator reaches its maximum after 4 hours. The dosage affects the duration of action.
During studies of the movement of blood through the vessels in people with primary hypertension, a decrease in blood pressure was accompanied by insignificant changes in heart rate indicators and a reduction in general parasympathetic obstruction vessels.
Enalapril treatment:
- leads to a significant regression of the pathology of the left ventricle and stabilization of its contractile ability;
- positively affects the ratio of risk factors for the development of cardiovascular diseases in the blood;
- has no effect, or has a positive effect on the level of total cholesterol.
After oral administration, the substance is intensively absorbed in the digestive system. Within 1 hour, the concentration in serum plasma reaches a maximum. The degree of absorption is approximately 60%. The substance undergoes intensive hydrolysis, forming the substances of enalaprilat. Already after 3 hours after oral administration, its content in the blood reaches its maximum values.
Excretion is carried out through the kidneys. Metabolites detected in urine: approximately 40% of enalaprilat and enalapril in their original form. The half-life of the active component during the course of drug treatment is carried out in 11 hours. Absorption does not depend on the diet.
Hydrochlorothiazide
This diuretic and antihypertensive substance does not affect the optimal blood pressure parameters. Stimulates the release of chloride and sodium in approximately equal volumes. The release of urine after oral administration occurs within 2 hours, within 4 hours the maximum effect is achieved and lasts from 6 to 12 hours.
The substance is not metabolized, however, it is rapidly excreted by the kidneys. In the case of monitoring the concentration of a substance in the blood during the day, the half-life is 5-15 hours. At least 61% of the dose taken is excreted unchanged throughout the day.
Combination of both substances
Thanks to clinical studies, it became clear that the use of a combination of these components provokes significant lowering blood pressure in comparison with therapy separately with each component and retains the effect at least for days. Korenitek is a convenient drug form for a course of treatment with a combination of both substances, which practically does not affect the bioavailability of each.
Indications for use
Korenitek (indications for use are due to the drug effect) is intended for the treatment of mild to moderate hypertension arteries in patients whose condition is normalized by using the active components of the drug separately in identical dosages.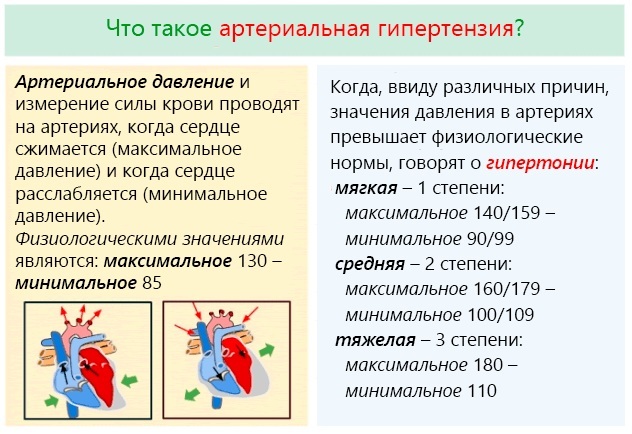
Contraindications
Korenitek is not recommended to be used for therapy when diagnosing the following pathologies:
- Anuria.
- Intolerance to any of the components of the drug.
- The presence of hereditary or idiopathic angioedema, or triggered by therapy with ACE inhibitors.
- Complex treatment with aliskiren, or drugs containing this component in diabetics and people suffering from kidney disorders.
- Children under the age of majority.
- Severe disorders of the liver and kidneys.
- Hereditary lactose intolerance, lactase deficiency.
Recommended appointment with precaution for pathologies:
- narrowing of the arteries of the kidneys;
- abdominal or mitral vasoconstriction;
- liver disorder syndrome;
- discirculatory encephalopathy or ischemia of the heart;
- systemic connective tissue pathologies;
- period after kidney transplant;
- disorders of hematopoiesis in the bone marrow system;
- diabetes;
- elevated plasma potassium;
- conditions characterized by a decrease in the volume of circulating plasma;
- postoperative period, or the use of general anesthesia.
ACE inhibitors are not recommended to be prescribed when carrying a child in the first trimester, and in the second and third trimester it is completely prohibited. If treatment with these medications is necessary, women planning to conceive are prescribed alternative antihypertensive therapy that is safe for the expectant mother and child. When conception has taken place, the use is immediately discontinued and alternative medications are prescribed.
Method of administration and dosage
Korenitek (indications for use imply oral administration of the medication) must be taken according to the instructions for it.
Arterial hypertension
The recommended daily dosage is 1 tablet. The dose, if indicated, can be increased to 2 tablets.
Pretreatment with diuretics
The first reception of Korenitek is able to provoke the onset of symptomatic hypertension. This symptom most often appears in patients with disorders of water and electrolyte balance, developed against the background of prior treatment with diuretic medications. Reception of the latter is stopped no later than 2 days before the first dose of Korenitek.
Kidney function disorders
In people suffering from these pathologies, thiazides may not give a sufficient effect. In this situation, taking the medication in monotherapy begins with 5-10 mg.
Elderly people
Clinical trials have shown the safety and efficacy of complex drug treatment in patients with hypertension, regardless of age. Adjustment of the primary dosage for this category of patients is not necessary.
Side effects
According to the tests carried out, the negative reactions that occur against the background of the use of the medication are of a moderate nature. Cancellation of treatment is not required.
The following side effects may occur:
| Systems and organs | Possible negative reactions |
| Respiratory | Cough, shortness of breath. |
| Cardiovascular | Feeling of palpitations, loss of consciousness, a strong decrease in blood pressure, tachycardia, pain in the sternum. |
| Bones and muscles | Muscle cramps, joint pain. |
| Kidney | Disorders of the functioning of the organ. |
| CNS | Dizziness, overexcitement, chronic fatigue, drowsiness, excessive fatigue, numbness, sleep disorders, migraine. |
| Digestive | Indigestion, abdominal pain, nausea, indigestion, vomiting, pancreatitis, stool disorders, a feeling of dryness in the mouth. |
| Allergy | Rash, itching. |
| Organs of the reproductive system | Decreased sex drive, development of impotence. |
| Others | Gout, tinnitus. |
It is extremely rare that a medication can provoke the development of the following negative consequences: angioedema of the lips, face, glottis, tongue, throat, extremities, hyperhidrosis, Quincke's edema, hyperhidrosis.
Overdose
When an excessive amount of enalapril is used, a significant drop in blood pressure (after about 6 hours) and numbness can be observed. An overdose of hydrochlorothiazide is accompanied by excessive diuresis, which provokes dehydration.
special instructions
With a low concentration of sodium in plasma, severe disorders of the functioning of the heart muscle, pathologies of the left ventricle, after the first use of the medication hypotension with appropriate clinical symptoms may occur, especially in patients with a decrease in circulating blood volume caused by diuretic therapy means. This condition is not a reason to cancel medication treatment.
It is important to constantly monitor the amount of serum electrolytes in order to detect possible imbalances in order to correct the indicators in a timely manner. This activity is necessary for patients with prolonged diarrhea and vomiting.
Treatment with this medication requires control of the following symptoms: thirst, a feeling of dryness in the mouth, digestive disorders, weakness, oliguria, drowsiness, excessive excitability, tachycardia, aches, hypotension and convulsions. When the angioteca of the face is formed, it is enough to cancel therapy and take antihistamines.
The defeat of angioedema of the pharynx, tongue or larynx can provoke death. With the development of this disorder, it is important to immediately administer epinephrine and, through intubation and tracheostomy, maintain airway patency.
In patients taking thiazide diuretics, intolerance reactions can be observed both in those who are prone to allergic reactions and in those who do not have such a problem. There is evidence of a worsening condition in people diagnosed with systemic lupus erythematosus.
This medication is not recommended to be prescribed to patients on hemodialysis, since the risk of anaphylactic reactions is quite high.
Before surgery (as well as dental manipulations), it is necessary to notify the anesthesiologist about the medication intake. In the course of treatment, there is a dry, prolonged cough that passes after the completion of the medication.
For the first time days of taking this medication, drowsiness, a strong decrease in blood pressure and dizziness may occur, which may adversely affect the ability to drive and engage in activities that may constitute danger. Therefore, during this period, driving and work that requires increased concentration and reaction speed is better to exclude.
Drug interactions
Korenitek (indications for use indicate the possibility of joint administration with other antihypertensive drugs, in this case, there is a total action) in complex therapy with potassium-containing diuretics and salts, potassium supplements, significantly increases the likelihood of developing hyperklemia.
When combined with lithium medications, the excretion of lithium through the kidneys is reduced, which increases the likelihood of developing intoxication with this substance.
NSAIDs reduce the effectiveness of the medication. Such a combination for patients with kidney dysfunction can provoke a deterioration in the functioning of the organ. However, these violations can be eliminated.
Ethanol and estrogens reduce the antihypertensive effect of the drug.
Co-administration with cytostatics, immunosuppressants and Allopurinol increases the risk of dysfunction of blood cells, or its cellular composition.
Analogs
The following medicines are analogs of Korenitek:
- Enalapril;
- Enap-N;
- Enalozide;
- Berlipril Plus;
- Enapril-N.
Storage conditions
Store Korenitic at a temperature not exceeding +30 C, in a place where there is no access to small children. The shelf life of tablets packaged in blisters is 3 years, and in high-density bottles - 2 years. The main indication is hypertension. It is strictly contraindicated for use at the end of the expiration date indicated on the package.
Video about the action of the drug Co-Rentitek
Quickly about hydrochlorothiazide and enalapril: