It is generally accepted that all elements of the chemical table that have permissible values do not pose a threat to humans. But this figure is negligible. For example, an increased level heavy metal salts has a detrimental effect on the body. This problem occurs frequently. The main provocateur is environmental problems.
Record content:
-
1 Types and composition
- 1.1 Mercury
- 1.2 Lead
- 1.3 Arsenic
- 1.4 Cadmium
- 1.5 Bismuth
- 1.6 Copper
- 1.7 Nickel
- 1.8 Antimony
- 1.9 Taliy
- 1.10 Cobalt
- 1.11 Manganese
- 1.12 Chromium
- 2 How they appear in the body
- 3 Impact
- 4 Beneficial features
- 5 Harmful properties
- 6 Side effects
- 7 Products table
- 8 Video about heavy metal salts
Types and composition
Heavy metal salts are substances that contribute to environmental pollution. The elements have a negative impact on health. For professional purposes, they are used in the medical and metallurgical field. In nature, there are about 40 species. However, not all are found in everyday life.
Mercury
It is contained in a small amount in the earth's crust: the indicator is 10-6% (mass). It is very rarely found as a nugget interspersed with rocks. In nature, it is represented as mercury sulfide HgS of a bright red color.

It is the only metal that can be liquid at room temperature. The simple substance is silver colored. The metal is easy to melt. The density is 13.55 g / cm 3.
Lead
Lead is a heavy and dense metal that has a bluish gray color. On contact with air, it loses its shine and becomes covered with an oxide-based film. Lead occurs frequently in nature and can be easily mined, which explains its long-standing fame. Keeps softness at a high density. It is easily scratched at a temperature of 20 ° C.
Arsenic
Arsenic is fragile. The metal is painted gray. It has a rhombohedral crystal lattice. When heated to 600 ° C, it sublimes. After cooling the vapors, a new compound is formed - yellow arsenic. At temperatures above 270 ° C, all As forms are converted to black arsenic.
Cadmium
Cadmium has similar properties to zinc. Usually found as an impurity in zinc ores.  The content index in the earth's crust is about 10-5% (mass.). The metal is painted silver-white. Differs in softness, ductility and ductility. During hydrolysis, salts are acidic.
The content index in the earth's crust is about 10-5% (mass.). The metal is painted silver-white. Differs in softness, ductility and ductility. During hydrolysis, salts are acidic.
Bismuth
The element is rare in nature. The content in the earth's crust is 0.00002% (mass.). In nature, it can be found both in a free state and in the form of compounds. In the free state, the element is colored pinkish-white. The density index is 9.8 g / cm 3. Metal is fragile. It does not oxidize when exposed to air and at room temperature.
Copper
Pure copper is a metal that characterizes ductility and toughness. Easily rolled into thin sheets. It is a conductor of heat and electric current. When the air is dry, it does not change its property, since the oxide film formed on the surface serves as a barrier against oxidation. When exposed to water and carbon dioxide, it becomes coated with a green bloom.
Nickel
The metal has a silvery color and a yellow tint. Attracted by a magnet. It is resistant to corrosion, air, water and alkalis. Chemical resistance is due to the ability to form oxide films.
Antimony
Usually in nature, antimony is found in combination with sulfur. The content index in the earth's crust is 0.00005% (mass.)  Antimony is a silvery white crystals with luster. The density index is 6.68 g / cm 3. The crystalline element is brittle and has poor heat and electricity conductivity.
Antimony is a silvery white crystals with luster. The density index is 6.68 g / cm 3. The crystalline element is brittle and has poor heat and electricity conductivity.
Taliy
A bluish white metal that is poisonous. The element is characterized by softness and rapid oxidation when released into the atmosphere. The content in the air should not be higher than 0.004 mg / m. For water, a dangerous indicator is 0.0001 mg / m. In nature, these values are usually not exceeded.
Cobalt
Cobalt is extremely rare in nature. The content in the earth's crust is 0.004% (mass.). Most often, the metal is combined with arsenic. It is distinguished by its hardness and ductility.
Manganese
Manganese is a commonly found element. It makes up 0.1% (mass.) Of the earth's crust.  It is a brittle and hard silver-colored metal. The density index is 7.44 g / cm 3, and the melting point is 1245 ° C.
It is a brittle and hard silver-colored metal. The density index is 7.44 g / cm 3, and the melting point is 1245 ° C.
Chromium
Metal characterizes hardness. It is gray-colored, shiny and polished.  Used in stainless steel alloys. The human body needs a low amount of chromium to metabolize sugar. Chromium is highly toxic.
Used in stainless steel alloys. The human body needs a low amount of chromium to metabolize sugar. Chromium is highly toxic.
How they appear in the body
Heavy metal salts are substances that people living in large cities and industrial centers are exposed to. It is in these areas that an increased concentration of substances is recorded. Can enter the body with inhaled air, food and water.
Insufficiently treated wastewater from factories and plants contains a high concentration of chemical compounds. The most common source of poisoning is fish and seafood. The phenomenon is explained by the pollution of the World Ocean. Substances can be found in products grown on disadvantaged land.
Many substances are included in the composition of pesticides used in agriculture. For example, fungicides including copper are used. They are used to prevent the growth of fungi.
Metal salts can be found in the following foods:
- animal meat;
- milk;
- cereals;
- fruits;
- vegetables.
May also be a part of:
- medicines;
- tobacco smoke;
- paints;
- exhaust gases;
- petroleum products;
- cans.
Along the food chain, they are transmitted to the human and animal body. Substances from the soil penetrate into plant roots. Mushrooms have a high level of accumulation. A mycelium with a branched root system absorbs toxins like a sponge. Therefore, the edible mushrooms growing along the highway are classified as poisonous.
The main method of penetration into the body is the gastrointestinal tract. A sign of poisoning is a metallic taste in the mouth. In some cases, the smell of acetone appears.
Impact
With an increased marker in the child's body, serious pathologies are provoked. There is a violation of the functionality of the central nervous system.
It often manifests itself:
- migraine;
- increased level of fatigue;
- irritability;
- delayed intellectual development;
- tremor of the limbs.
Long-term exposure to arsenic on a pregnant woman provokes a defect in the intrauterine development of the fetus, the development of leukemia cancer in the child, pathology of the respiratory organs and the genitourinary system.
When heavy metal salts enter the body of adults, the following symptoms are noted:
- failure of the cardiovascular system; neurological pathology;
- intense migraine;
- deterioration in attention and memorization;
- insomnia;
- hyperhidrosis;
- anemia;
- violation of bone morphology;
- redness of the skin;
- swelling;
- hyperemia;
- inflammation of the mucous membranes;
- sharp pain in the epigastric region;
- nausea;
- diarrhea.
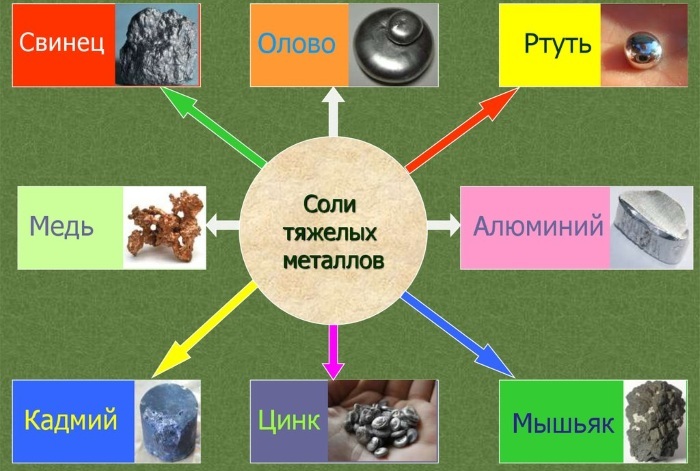
Heavy metal salts.
Chronic intoxication causes:
- asthenia;
- violation of the immune system;
- deterioration in general health;
- temperature increase.
Poisoning with lead and mercury is especially dangerous. It provokes damage to the tooth enamel and intense pain in the oral cavity. Bleeding of the gums and excessive salivation may also occur.
An inflammatory process is provoked in the larynx, trachea and nasal cavity. The lymph nodes are involved in the inflammatory process. A sign of poisoning is the appearance of blue impurities in the vomit.
The table shows the effect of substances on organs and systems:
| Organs and systems | Symptoms |
| Gastrointestinal tract | In severe poisoning, constipation is provoked. In some cases, bleeding is observed. In the presence of a stomach ulcer, blood loss can be significant. As a rule, the pathology begins to progress. When metal salts enter the esophagus, a person experiences heartburn and burning. |
| The cardiovascular system | A person may complain of a sharp fluctuation in blood pressure and bradycardia. Jumps in blood pressure can provoke the development of stroke and heart attack. A person has shortness of breath, cyanosis and hyperemia. Some skin areas become pale, depending on the level of pigment, circulation and hemoglobin marker. |
| Nervous system | There are memory problems, weakness, apathy. Occasionally, convulsive seizures, difficulty in movement and lack of coordination are traced. A person may be euphoric or aggressive, experience insomnia and phobias. In some cases, hallucinations, disorientation in space and psychomotor agitation develop. The most severe forms include stopper, loss of consciousness and coma. |
| Respiratory system | There is soreness in the chest, dry, unpleasant cough mixed with blood, and irritation of the respiratory tract. Dermatitis and allergic reactions are often provoked. |
| Kidney and liver | The organs are under enormous stress. At the initial stages, a decrease in daily urine output is formed. Urine often turns green. Blood biochemical markers change, especially laboratory values for creatine and urea. |
| Eyes | All types of poisoning are inherent in visual disturbances. A person has diplopia, a decrease or loss of vision. |
| Blood | A dangerous sign is blood hemolysis, which is characterized by the destruction of red blood cells and the release of hemoglobin into the blood. As a result, the blood turns blue. The skin also has a bluish tint. |
Beneficial features
Heavy metal salts are substances that take part in biological processes. A certain amount of trace elements is needed for the functionality of plants, animals and humans.
Substances that have an antimicrobial effect deactivate the enzymes necessary for the vital activity of bacteria. Enzyme inactivation occurs when metal ions interact with sulfhydryl groups (SH-groups). Salt has a local effect on the skin and mucous membranes.
Ions are used as antiseptics:
- mercury (dichloride);
- silver (protargol);

- zinc (zinc sulfate);
- bismuth (dermatol, xeroform).
When used as an antiseptic, it should be remembered that the level of antimicrobial exposure is reduced in an environment with a high protein content (for example, it is contained in pus or blood). In this case, substances for disinfection become unsuitable.
Harmful properties
Heavy metal salts can be both beneficial and harmful. Substances have more negative characteristics. The danger lies in the ability to destroy cell membranes and change the degree of their permeability.
This entails a violation of the structure of the nucleic acid and a change in the functional and qualitative parameters of the protein. The processes form spontaneous mutations and disrupt the metabolism in the body, which leads to the failure of all organs and systems. The compounds carry a high risk of developing cancer.
Side effects
The table shows the harmful properties of metals.
| Metal type | Specific symptoms |
| Lead | It provokes a failure of the functionality of the central nervous system. Poisoning has an extremely negative effect on the female body. In particular, the reproductive system suffers.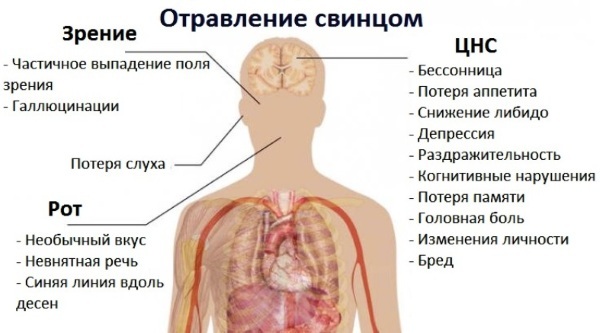 There are constant painful sensations in the bones, nausea and urge to vomit in the morning, weight loss. The body temperature indicator drops sharply. Toxic shock is provoked, which can be fatal. |
| Copper | Negatively affects the kidneys, liver and nervous system. |
| Mercury vapor | Violate the functionality of the respiratory system. They affect the nervous and cardiovascular system. |
| Antimony | It causes a decrease in appetite, provokes itching and inflammation of the oral cavity, affects the genitourinary system of women. |
| Thallium | It provokes baldness, muscle weakness and cardiovascular disorders. Affects the bronchi and lungs. Often, poisoning provokes death.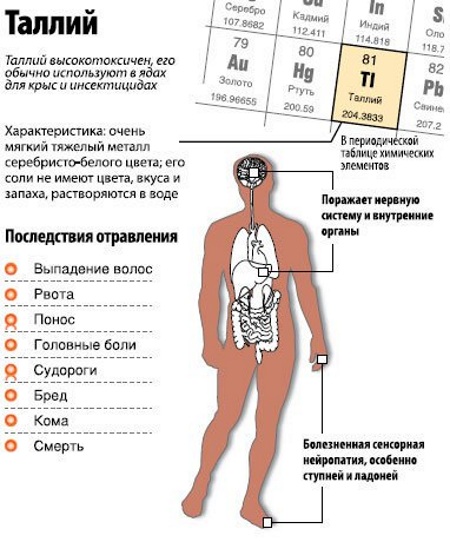
|
| Arsenic | Metal salts cause systemic damage to the body. |
| Cobalt | Provokes hearing loss. |
| Cadmium | Reduces appetite, disrupts the absorption of the renal tubules, destroys the bone and muscular system. The defeat of internal organs is systemic. |
| Zinc | Promotes the development of anemia and hearing loss. |
| Chromium | Weakens the immune system, impairs absorption. Provokes the formation of a through hole in the cartilage of the nasal septum. |
| Manganese | Provokes asthenia. Causes depressive disorders and asthenia. |
A huge role in heavy metal poisoning in humans is played by the way salts enter the body.
For example, when inhaling mercury vapor, it is provoked:
- cough;
- discharge of blood;
- temperature increase;
- pneumonia.
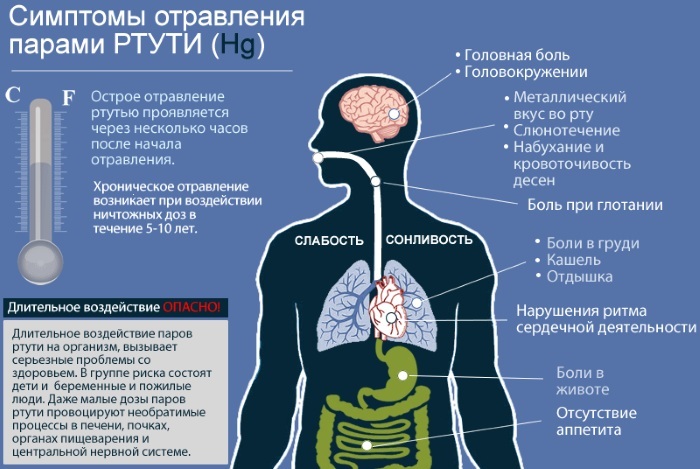
Dermatitis is common when exposed to the skin. The affected areas become red, blistered, and itchy. At a later stage, with deep penetration of salts into the layers of the skin, systemic disorders are provoked.
Products table
Heavy metal salts are substances found in foods. The table shows the main markers.
| Products | Lead | Cadmium | Arsenic | Mercury | Copper | Zinc |
| Beans and cereals | 0,5 | 0,1 | 0,2-0,3 | 0,02-0,03 | 10 | 50 |
| Sweets | 1,0 | 0,1 | 0,5 | 0,02-0,03 | 10-20 | 50 |
| Milk | 0,1 | 0,03 | 0,05 | 0,005 | 1,0 | 5 |
| Sunflower oil | 0,1 | 0,05 | 0,1 | 0,05 | 1,0 | 5-10 |
| Vegetables and fruits | 0,04-0,5 | 0,03 | 0,2 | 0,02 | 5,0 | 10,0 |
| Canned vegetables and fruits | 1,0 | 0,05 | 0,2 | 0,02 | 5,0 | 10,0 |
| Meat products | 0,5 | 0,05 | 0,1 | 0,03 | 5,0 | 20 |
| Canned meat products | 1,0 | 0,1 | 0,1 | 0,03 | 5,0 | 70 |
| Frozen and fresh fish | 1,0 | 0,2 | 1,0-5,0 | 0,3-0,6 | 10 | 40 |
| Canned fish | 1,0 | 0,2 | 1,0-5,0 | 0,3-0,7 | 10 | 40 |
When preparing and storing food, use dishes. As for plastic bags and plastic dishes, it is not recommended to store food in them for a long time.
The possibility of substance poisoning is advised to be taken seriously. Chronic intoxication is often diagnosed. It can be an occupational disease in people whose employment is associated with harmful conditions. The development of acute and chronic forms is not excluded under ordinary living conditions.
In order to remove the salts of heavy metals from their body, dairy products rich in calcium, foods with fiber content, dried fruits and cereals should be consumed more often. This diet promotes the elimination of substances from the body.
Video about heavy metal salts
Heavy metals as causative agents of various diseases:



